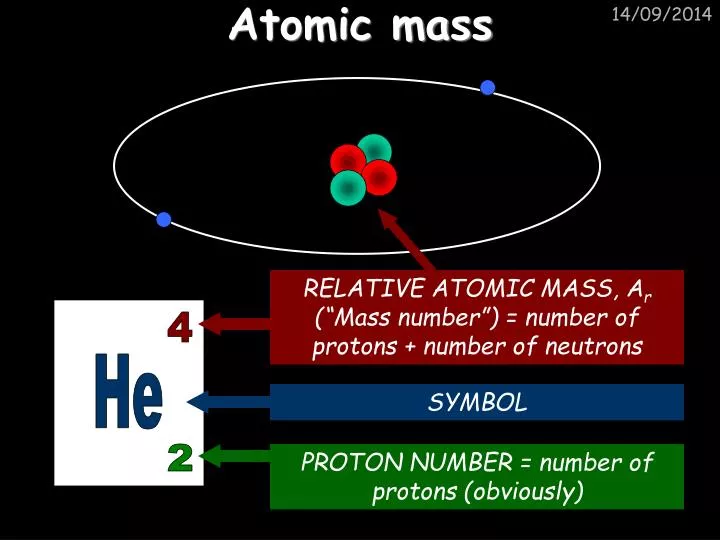
2 Locate atomic mass on the periodic table. This is a very useful property when it comes to practical calculations, as it allows easy conversion between the mass and moles of a given quantity of atoms or molecules of the same type. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. The atomic mass is the number of grams of the element in one mole of atoms of the element. Each atom of an element contains the same number of protons, known as the atomic number (Z). Please note that the elements do not show their natural relation towards each other as in the Periodic system. The mass of an atom is a weighted average that is largely determined by the number of its protons and neutrons, whereas the number of protons and electrons determines its charge. These are calculated by adding the relative atomic masses of the constituent elements, as given in the. The second isotope has an atomic mass of 36.96590 and has an abundance of 24.22. Relative molecular mass and relative formula mass. The first isotope has an atomic mass of 34.96885 and has an abundance of 75.78. 
The unity for atomic mass is gram per mol. Example: Calculating the atomic mass of a given chlorine sample where two isotopes are mixed. Atomic mass value sometimes change over time in publications as scientists revise the natural isotope abundance of elements. 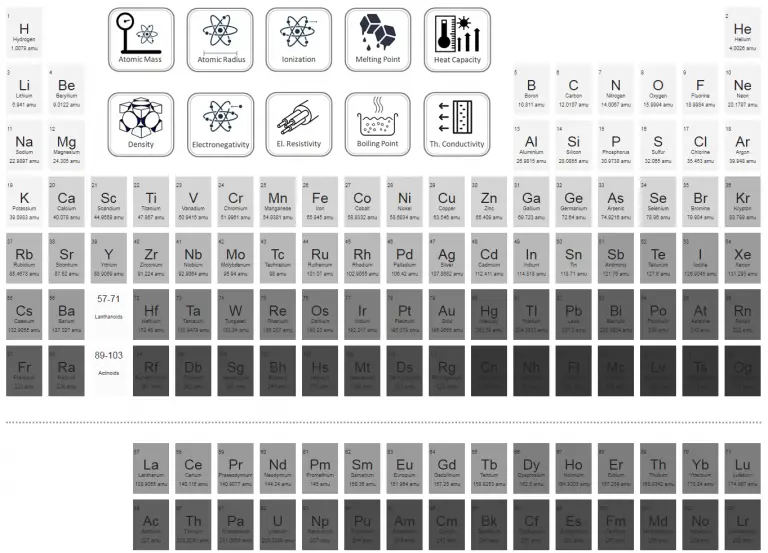
The atomic mass is the average number of protons and neutrons for all natural isotopes of an element. No single carbon atom has a mass of 12.01 amu, but in a handful of C atoms the average mass of the carbon atoms is. The mass number is the sum of the number of protons and neutrons in an atom. The lightest chemical element is Hydrogen and the heaviest is Hassium. This is the average atomic mass of carbon. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). The Atomic Mass Evaluation (AME) is the most reliable source for comprehensive information related to atomic masses. This list contains the 118 elements of chemistry. The atomic masses are important input parameters for nuclear astrophysics calculations. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects.Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
